GW Pharmaceuticals: Friend or Foe?

We all know that big pharma is the enemy: they shove harmful prescriptions down our throat. They block the legalization of cannabis. They operate a well-oiled marketing machine that keeps people sick. And some, like GW Pharmaceuticals, even worked with notorious killers, specifically one named Monsanto.
But now that GW Pharmaceuticals is creating cannabis-based medicine, are they helping? Maybe they just want more money to grow on trees.
Earlier this week, GW Pharmaceuticals announced that their experiments with cannabis have been a success (are we really surprised?). Over the past few years, they’ve been testing the efficacy of a cannabis-based medicine in treating glioblastoma multiforme, a form of brain cancer. The medicine was even shown to reduce the mortality rate. Patients with glioblastoma multiforme don’t have a very good outlook, as this cancer normally kills victims within 2 years. However, GW Pharmaceuticals said that their findings have yielded good news for patients.
In a statement, CEO Justin Gover said "We believe that the signals of efficacy demonstrated in this study further reinforce the potential role of cannabinoids in the field of oncology and provide GW with the prospect of a new and distinct cannabinoid product candidate in the treatment of glioma.”
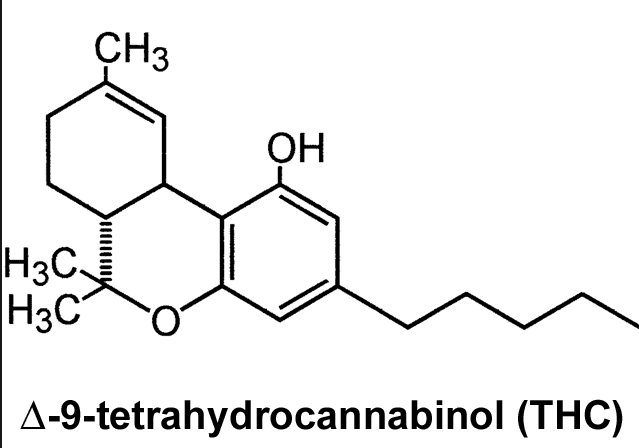
In the most recent trial phase, the patients were administered with both CBD and THC, as well as Temozolomide which is an oral chemotherapy medicine. Patients who used all three showed a 83% one-year survival rate, while the other participants who were given placebo only demonstrated a 53% survival rate. The combination of medications were granted an Orphan Drug Designation by the US FDA as well as the European Medicines Agency (EMA) in the treatment of glioblastoma multiforme
There is another positive way to take these news: the fact that such a big player in the pharmaceutical market would make cannabis-based medicine means simply that it does work. So it’s time for the feds and the DEA to take it seriously - and reschedule cannabis!
GW’s Cannabis-Based Spray For Epilepsy
That isn’t the only project in the pipeline - GW is definitely very interested in working with cannabis. Their cannabis projects have also increased the stock value of the company.
Last year, GW released data about the success of Epidolex, a CBD-based spray in treating 2 rare forms of pediatric epilepsy:
|
Disease |
Placebo (Reduction) in Seizure Frequency |
Epidiolex (Reduction) in Monthly Seizure Frequency |
|
Dravet Syndrome |
(13%) |
(39%) |
|
Lennox-Gastaut Syndrome-Trial 1 |
(22%) |
(44%) |
|
Lennox-Gastaut Syndrome-Trial 2 |
(17%) |
(42%) |
DATA SOURCE: GW PHARMACEUTICS.
The results showed very promising results in the efficacy of Epidolex. Even in lower doses, Epidolex worked just as well as a second dose for the second Lennox-Gastaut syndrome trial.
GW Pharma is set to apply for FDA approval before June. They also intend to submit a plan to regulators in Europe during the second half of 2017.
GW Gets Special Treatment For Sativex
GW’s other cannabis-based product, Sativex, has already been approved for the treatment of spasticity caused by multiple sclerosis. Sativex is already available in 16 countries outside of the USA including Spain, Australia, Canada, and the United Kingdom. It also recently just got approval to sell in Brazil, under the name Mevatyl.
So why does a company like GW Pharma get to make money out of a cannabis-based drug like Sativex, while the rest of the businesses in the industry have to suffer with numerous obstacles that have to do with regulation, banking, and many other headaches?
Despite sounding like a medicine, Sativex is an extract made from the cannabis plant. GW Pharma has been open about the fact that Sativex already contains the very same compounds that are already found in the cannabis plant. It doesn’t sound fair that GW Pharma has access to resources that allow the plant to be standardized for use in medicine - this means that each bottle of Sativex contains an equal 1:1 ratio of both CBD and THC. The absence of standardization has been an obstacle for the medical marijuana industry, unfortunately.
It’s also a little strange that Sativex is actually manufactured in the United Kingdom - a country where cannabis is ILLEGAL even for medical purposes. Despite this, in 1998 the government granted GW Pharmaceuticals a license to grow the plant for research and development purposes.
While this is happening, groups continue to lobby and work towards legalization in our very own shores where obtaining a pure form of cannabis as medicine is almost impossible for millions of sick patients. Smells like a monopoly to me.

On the other hand, there is another positive takeaway to these news: the fact that such a big player in the pharmaceutical market would make cannabis-based medicine means simply that it does work. This also means that people will be able to enjoy the medicinal properties of cannabis, BUT the only way to do so is by going through Big Pharma and giving them your money.
I’d prefer that my money go to the honest and hardworking people cultivating the plant and selling it to me at dispensaries, wouldn’t you?
What are your thoughts on GW Pharma working with cannabis? Share with us in the comments below!
OTHER ARTICLES YOU MAY ENJOY...
BIG PHARMA AND CANNABIS, CLICK HERE.







